
ACCELERATING
Clinical Rx Development
and Diagnostic Research toward Commercialization
DIAGNOSTICS PORTFOLIO
(INCELLKINE ASSAY)
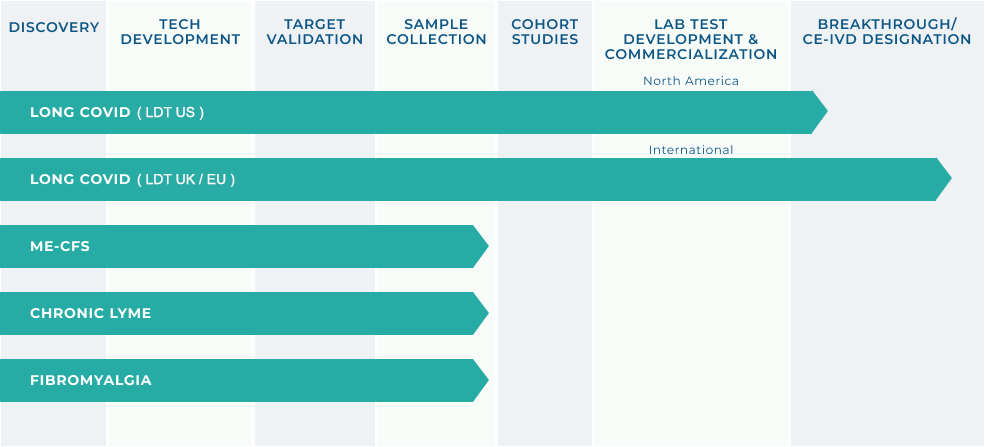
Lab partners located in USA and Globally to process patient testing
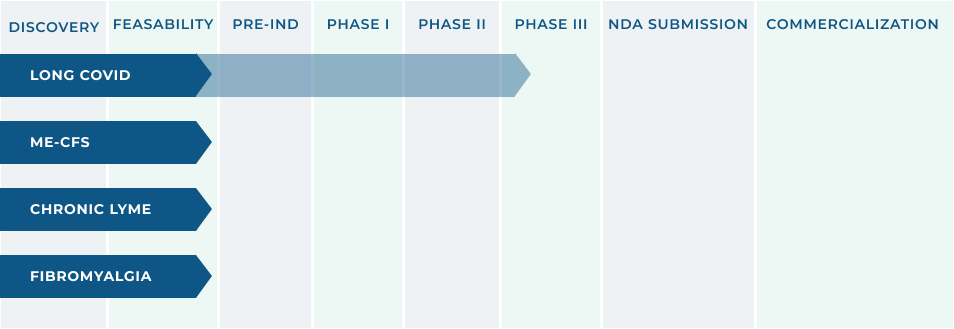
RX THERAPEUTIC INNOVATION
In January, 2024, our application was accepted by the FDA to proceed with a Randomized Clinical Trial for the Treatment of Long COVID/Post Acute Sequelae of COVID (PASC) with Selzentry® (maraviroc) in Combination with Lipitor (atorvastatin). The clinical trial will support a New Drug Application (NDA) filing through the 505(B)(2) pathway of the Federal Food, Drug, and Cosmetic Act (FDCA).
The initial focus is on the diagnosis and treatment of Long Covid with ongoing evaluation of other indications that present significant unmet medical needs.
BIOMETRIC REMOTE MONITORING

An integral personal device for your immune health and wellness
Biometric wearable helps patients track symptoms
Learn More

WATER & SHOCK RESISTANT
5 ATM Water and Shock Resistant
30 DAY DATA PERIOD
A month of health data memory
FCC & IC COMPLIANT
This equipment has been tested and found to comply with the limits for a Class B digital device, pursuant to Part 15 of the FCC Rules
LONG-LASTING BATTERY
7-10 day long-lasting battery life
VITAL HEALTH TRACKING
Tracks multiple vital health functions and activities important to managing your immunologic symptoms
CONTINOUS TRACKING
Allows uninterrupted tracking for longer intervals for more accurate tracking
Data that everyone can understand
REMOTE MONITORING DEVICE
Giving Patients Confidence
in Monitoring Their Health
Allows input of your symptoms and severity, laboratory test results, medications, supplements, and other daily information on your health status
Data can we shared with others to compare and discuss
iPhone and Android compatible
Compatible with Apple and Android watches
Reminder alerts for when to take your prescription medications, supplements, vitals and other important activities.
